This is “End-of-Chapter Material”, section 20.8 from the book Introduction to Chemistry: General, Organic, and Biological (v. 1.0). For details on it (including licensing), click here.
For more information on the source of this book, or why it is available for free, please see the project's home page. You can browse or download additional books there. To download a .zip file containing this book to use offline, simply click here.
20.8 End-of-Chapter Material
Chapter Summary
To ensure that you understand the material in this chapter, you should review the meanings of the bold terms in the following summary and ask yourself how they relate to the topics in the chapter.
Metabolism is the general term for all chemical reactions in living organisms. The two types of metabolism are catabolism—those reactions in which complex molecules (carbohydrates, lipids, and proteins) are broken down to simpler ones with the concomitant release of energy—and anabolism—those reactions that consume energy to build complex molecules. Metabolism is studied by looking at individual metabolic pathways, which are a series of biochemical reactions in which a given reactant is converted to a desired end product.
The oxidation of fuel molecules (primarily carbohydrates and lipids), a process called respiration, is the source of energy used by cells. Catabolic reactions release energy from food molecules and use some of that energy for the synthesis of adenosine triphosphate (ATP); anabolic reactions use the energy in ATP to create new compounds. Catabolism can be divided into three stages. In stage I, carbohydrates, lipids, and proteins are broken down into their individual monomer units—simple sugars, fatty acids, and amino acids, respectively. In stage II, these monomer units are broken down by specific metabolic pathways to form a common end product acetyl-coenzyme A (CoA). In stage III, acetyl-CoA is completely oxidized to form carbon dioxide and water, and ATP is produced.
The digestion of carbohydrates begins in the mouth as α-amylase breaks glycosidic linkages in carbohydrate molecules. Essentially no carbohydrate digestion occurs in the stomach, and food particles pass through to the small intestine, where α-amylase and intestinal enzymes convert complex carbohydrate molecules (starches) to monosaccharides. The monosaccharides then pass through the lining of the small intestine and into the bloodstream for transport to all body cells.
Protein digestion begins in the stomach as pepsinogen in gastric juice is converted to pepsin, the enzyme that hydrolyzes peptide bonds. The partially digested protein then passes to the small intestine, where the remainder of protein digestion takes place through the action of several enzymes. The resulting amino acids cross the intestinal wall into the blood and are carried to the liver.
Lipid digestion begins in the small intestine. Bile salts emulsify the lipid molecules, and then lipases hydrolyze them to fatty acids and monoglycerides. The hydrolysis products pass through the intestine and are repackaged for transport in the bloodstream.
In cells that are operating aerobically, acetyl-CoA produced in stage II of catabolism is oxidized to carbon dioxide. The citric acid cycle describes this oxidation, which takes place with the formation of the coenzymes reduced nicotinamide adenine dinucleotide (NADH) and reduced flavin adenine dinucleotide (FADH2). The sequence of reactions needed to oxidize these coenzymes and transfer the resulting electrons to oxygen is called the electron transport chain, or the respiratory chain. The compounds responsible for this series of oxidation-reduction reactions include proteins known as cytochromes, Fe·S proteins, and other molecules that ultimately result in the reduction of molecular oxygen to water. Every time a compound with two carbon atoms is oxidized in the citric acid cycle, a respiratory chain compound accepts the electrons lost in the oxidation (and so is reduced) and then passes them on to the next metabolite in the chain. The energy released by the electron transport chain is used to transport hydrogen (H+) ions from the mitochondrial matrix to the intermembrane space. The flow of H+ back through ATP synthase leads to the synthesis and release of ATP from adenosine diphosphate (ADP) and inorganic phosphate ions (Pi) in a process known as oxidative phosphorylation. Electron transport and oxidative phosphorylation are tightly coupled to each other. The enzymes and intermediates of the citric acid cycle, the electron transport chain, and oxidative phosphorylation are located in organelles called mitochondria.
The oxidation of carbohydrates is the source of over 50% of the energy used by cells. Glucose is oxidized to two molecules of pyruvate through a series of reactions known as glycolysis. Some of the energy released in these reactions is conserved by the formation of ATP from ADP. Glycolysis can be divided into two phases: phase I consists of the first five reactions and requires energy to “prime” the glucose molecule for phase II, the last five reactions in which ATP is produced through substrate-level phosphorylation.
The pyruvate produced by glycolysis has several possible fates, depending on the organism and whether or not oxygen is present. In animal cells, pyruvate can be further oxidized to acetyl-CoA and then to carbon dioxide (through the citric acid cycle) if oxygen supplies are sufficient. When oxygen supplies are insufficient, pyruvate is reduced to lactate. In yeast and other microorganisms, pyruvate is not converted to lactate in the absence of oxygen but instead is converted to ethanol and carbon dioxide.
The amount of ATP formed by the oxidation of glucose depends on whether or not oxygen is present. If oxygen is present, glucose is oxidized to carbon dioxide, and 36–38 ATP molecules are produced for each glucose molecule oxidized, using the combined pathways of glycolysis, the citric acid cycle, the electron transport chain, and oxidative phosphorylation. Thus, approximately 42% of the energy released by the complete oxidation of glucose is conserved by the synthesis of ATP. In the absence of oxygen, only 2 molecules of ATP are formed for each molecule of glucose converted to lactate (2 molecules), and the amount of energy conserved is much less (2%).
Fatty acids, released by the degradation of triglycerides and other lipids, are converted to fatty acyl-CoA, transported into the mitochondria, and oxidized by repeated cycling through a sequence of four reactions known as β-oxidation. In each round of β-oxidation, the fatty acyl-CoA is shortened by two carbon atoms as one molecule of acetyl-CoA is formed. The final round of β-oxidation, once the chain has been shortened to four carbon atoms, forms two molecules of acetyl-CoA. β-oxidation also forms the reduced coenzymes FADH2 and NADH, whose reoxidation through the electron transport chain and oxidative phosphorylation leads to the synthesis of ATP. The efficiency of fatty acid oxidation in the human body is approximately 41%.
Amino acids from the breakdown of proteins can be catabolized to provide energy. Amino acids whose carbon skeletons are converted to intermediates that can be converted to glucose through gluconeogenesis are known as glucogenic amino acids. Amino acids whose carbon skeletons are broken down to compounds used to form ketone bodies are known as ketogenic amino acids.
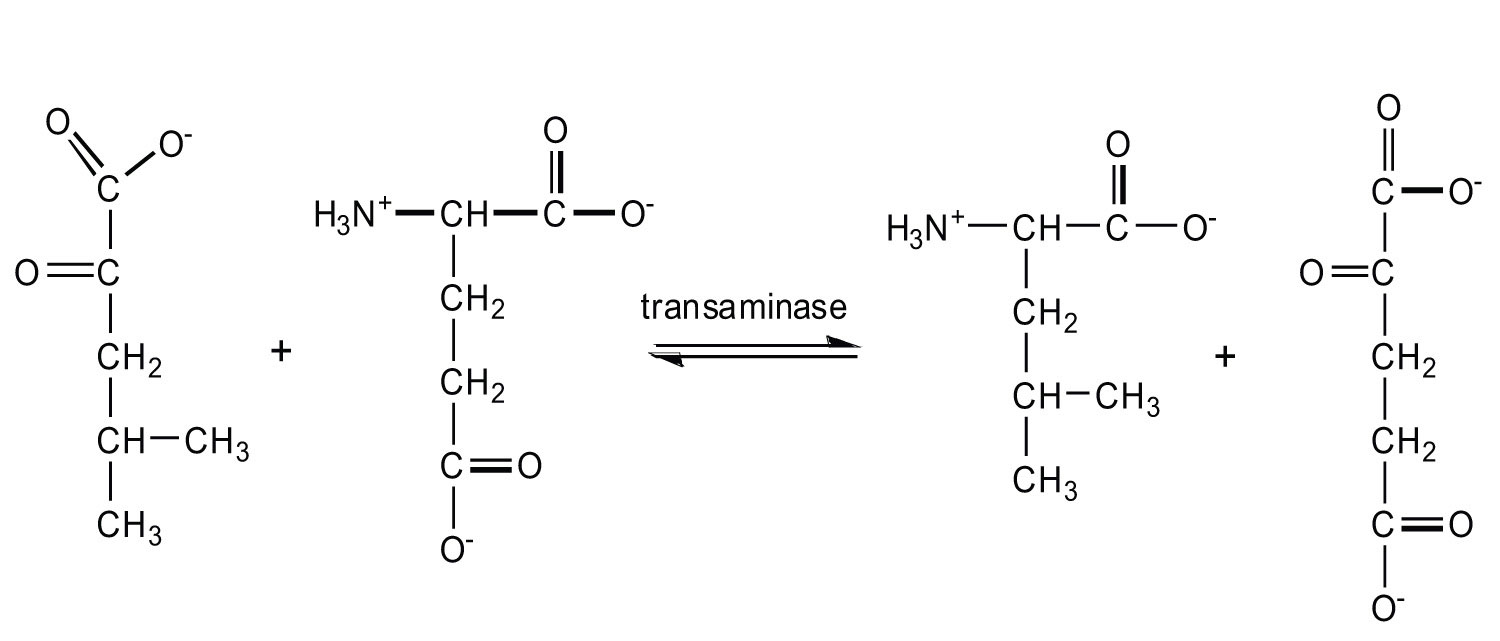
The first step in amino acid catabolism is separation of the amino group from the carbon skeleton. In a transamination, the amino acid gives its NH2 to pyruvate, α-ketoglutarate, or oxaloacetate. The products of this reaction are a new amino acid and an α-keto acid containing the carbon skeleton of the original amino acid. Pyruvate is transaminated to alanine, α-ketoglutarate to glutamate, and oxaloacetate to aspartate. The amino groups used to form alanine and aspartate are ultimately transferred to α-ketoglutarate, forming glutamate. The glutamate then undergoes oxidative deamination to yield α-ketoglutarate and ammonia.
Additional Exercises
-
Hydrolysis of which compound—arginine phosphate or glucose 6-phosphate—would provide enough energy for the phosphorylation of ATP? Why?
-
If a cracker, which is rich in starch, is chewed for a long time, it begins to develop a sweet, sugary taste. Why?
-
Indicate where each enzymes would cleave the short peptide ala-ser-met-val-phe-gly-cys-lys-asp-leu.
- aminopeptidase
- chymotrypsin
-
Indicate where each enzymes would cleave the short peptide ala-ser-met-val-phe-gly-cys-lys-asp-leu.
- trypsin
- carboxypeptidase
-
If the methyl carbon atom of acetyl-CoA is labeled, where does the label appear after the acetyl-CoA goes through one round of the citric acid cycle?
-
If the carbonyl carbon atom of acetyl-CoA is labeled, where does the label appear after the acetyl-CoA goes through one round of the citric acid cycle?
-
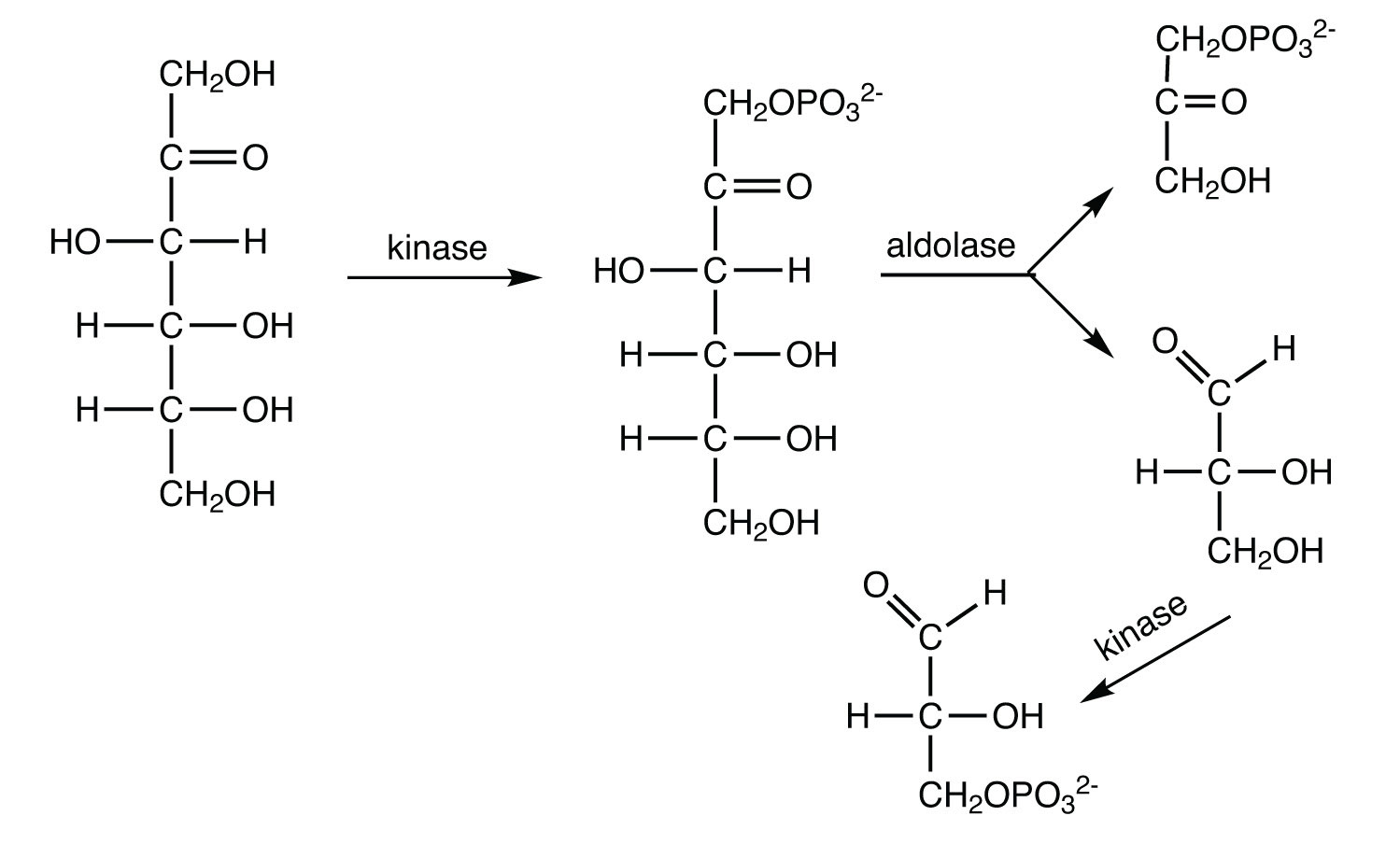
The average adult consumes about 65 g of fructose daily (either as the free sugar or from the breakdown of sucrose). In the liver, fructose is first phosphorylated to fructose 1-phosphate, which is then split into dihydroxyacetone phosphate and glyceraldehyde. Glyceraldehyde is then phosphorylated to glyceraldehyde 3-phosphate, with ATP as the phosphate group donor. Write the equations (using structural formulas) for these three steps. Indicate the type of enzyme that catalyzes each step.
-
What critical role is played by both BPG and PEP in glycolysis?
-
How is the NADH produced in glycolysis reoxidized when oxygen supplies are abundant?
-
When a triglyceride is hydrolyzed to form three fatty acids and glycerol, the glycerol can be converted to glycerol 3-phosphate and then oxidized to form dihydroxyacetone phosphate, an intermediate of glycolysis. (In this reaction, NAD+ is reduced to NADH.) If you assume that there is sufficient oxygen to completely oxidize the pyruvate formed from dihydroxyacetone phosphate, what is the maximum amount of ATP formed from the complete oxidation of 1 mol of glycerol?
-
How is the FADH2 from β-oxidation converted back to FAD?
-
If 1 mol of alanine is converted to pyruvate in a muscle cell (through transamination) and the pyruvate is then metabolized via the citric acid cycle, the electron transport chain, and oxidative phosphorylation, how many moles of ATP are produced?
-
If the essential amino acid leucine (2-amino-4-methylpentanoic acid) is lacking in the diet, an α-keto acid can substitute for it. Give the structure of the α-keto acid and the probable reaction used to form leucine from this α-keto acid.
Answers
-
The hydrolysis of arginine phosphate releases more energy than is needed for the synthesis of ATP, while hydrolysis of glucose 6-phosphate does not.
-
-
- The enzyme will cleave off amino acids one at a time beginning with alanine (the N-terminal end).
- following phenylalanine
-
-
Half of the label will be on the second carbon atom of oxaloacetate, while the other half will be on the third carbon atom.
-
-
-
-
When oxygen is abundant, NADH is reoxidized through the reactions of the electron transport chain.
-
-
FADH2 is reoxidized back to FAD via the electron transport chain.
-
-