This is “Chemical Properties of Alkenes”, section 13.4 from the book Introduction to Chemistry: General, Organic, and Biological (v. 1.0). For details on it (including licensing), click here.
For more information on the source of this book, or why it is available for free, please see the project's home page. You can browse or download additional books there. To download a .zip file containing this book to use offline, simply click here.
13.4 Chemical Properties of Alkenes
Learning Objective
- Write equations for the addition reactions of alkenes with hydrogen, halogens, and water.
Alkenes are valued mainly for addition reactionsA reaction in which substituent groups join to hydrocarbon molecules at points of unsaturation—the double or triple bonds., in which one of the bonds in the double bond is broken. Each of the carbon atoms in the bond can then attach another atom or group while remaining joined to each other by a single bond.
Perhaps the simplest addition reaction is hydrogenationA reaction in which hydrogen gas reacts at a carbon-to-carbon double or triple bond or a carbon-to-oxygen double bond to add hydrogen atoms to carbon atoms.—a reaction with hydrogen (H2) in the presence of a catalyst such as nickel (Ni) or platinum (Pt).
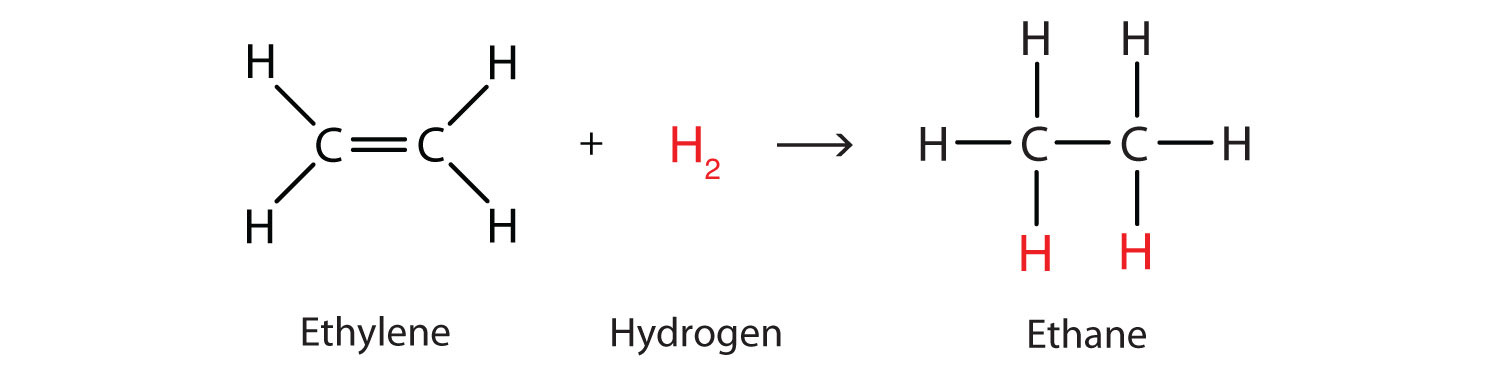
The product is an alkane having the same carbon skeleton as the alkene. (The use of hydrogenation to convert unsaturated vegetable oils to saturated fats is discussed in Chapter 17 "Lipids", Section 17.2 "Fats and Oils".)
Alkenes also readily undergo halogenationA reaction in which a halogen reacts at a carbon-to-carbon double or triple bond to add halogen atoms to carbon atoms.—the addition of halogens. Indeed, the reaction with bromine (Br2) can be used to test for alkenes. Bromine solutions are brownish red. When we add a Br2 solution to an alkene, the color of the solution disappears because the alkene reacts with the bromine:

Another important addition reaction is that between an alkene and water to form an alcohol. This reaction, called hydrationThe addition of water to a substance; in organic chemistry, the addition of water across the carbon-to-carbon double bond of an alkene or the carbon-to-oxygen double bond of an aldehyde or ketone., requires a catalyst—usually a strong acid, such as sulfuric acid (H2SO4):
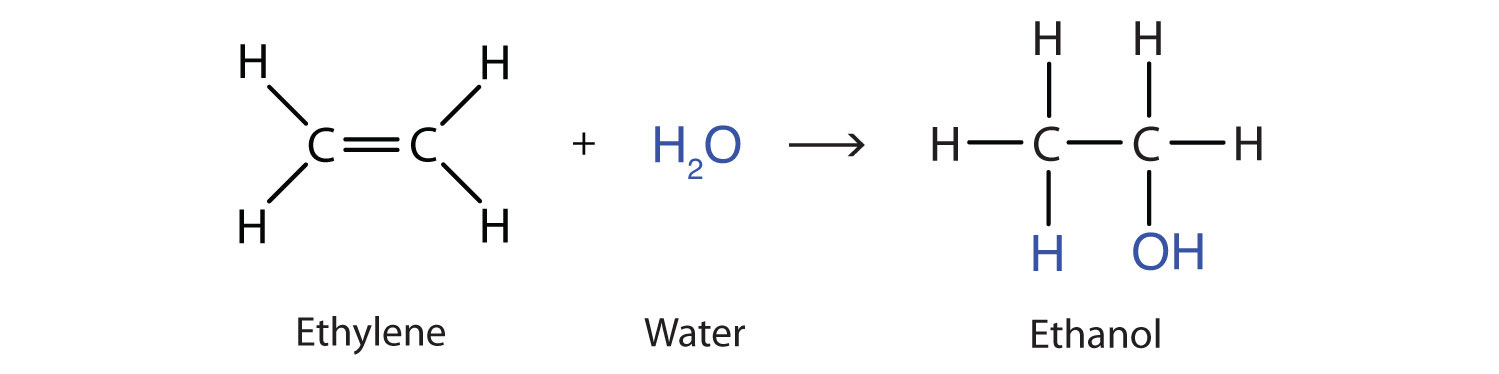
The hydration reaction is discussed further in Chapter 14 "Organic Compounds of Oxygen", Section 14.4 "Reactions That Form Alcohols", where we deal with this reaction in the synthesis of alcohols.
Example 4
Write the equation for the reaction between CH3CH=CHCH3 and each substance.
- H2 (Ni catalyst)
- Br2
- H2O (H2SO4 catalyst)
Solution
In each reaction, the reagent adds across the double bond.
Skill-Building Exercise
-
CH3CH2CH=CH2 with H2 (Ni catalyst)
-
CH3CH=CH2 with Cl2
-
CH3CH2CH=CHCH2CH3 with H2O (H2SO4 catalyst)
Write the equation for each reaction.
Concept Review Exercises
-
What is the principal difference in properties between alkenes and alkanes? How are they alike?
-
If C12H24 reacts with HBr in an addition reaction, what is the molecular formula of the product?
Answers
-
Alkenes undergo addition reactions; alkanes do not. Both burn.
-
C12H24Br2
Key Takeaway
- Alkenes undergo addition reactions, adding such substances as hydrogen, bromine, and water across the carbon-to-carbon double bond.
Exercises
-
Complete each equation.
- (CH3) 2C=CH2 + Br2 →
-

-
Complete each equation.
Answer
-
- (CH3)2CBrCH2Br
- CH3CH(CH3)CH2CH3
-

-








